Specialist MedTech consultancy
Want more than just a price and a tag line for your medical technology? Coreva Scientific builds value for your product. Our expertise is applied health economics and value-based healthcare. Whether it be pricing, cost effectiveness, burden of disease, or new value streams – Coreva Scientific are your partner from ideas to success. For more information , please have a look at our brochure.
Why work with a MedTech specialist?
“The Coreva service is excellent. There is nothing to improve. Everyone is courteous. The work is clean and professional. The dedication to the work is evident.”
– Client feedback, May 2021
A data-driven consultancy
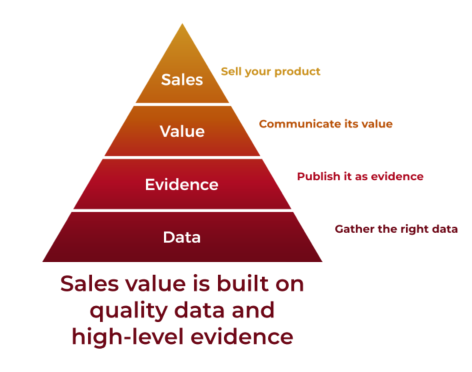
Coreva Scientific is a young and dynamic consultancy for the healthcare industry. Our skill lies in making health and economic data talk for your product. We help our partners develop new value for their products, or identify where their product can have the greatest impact on patients and healthcare providers. Interested in quantifying value? Want to determine return on investment? Need to collect the right health and economic data? Considering a meta-analysis? Coreva Scientific can help make the most of your data and your investment.
Product value creation
Customized data collection
Models
Market access
Data analysis
Medical device specialists
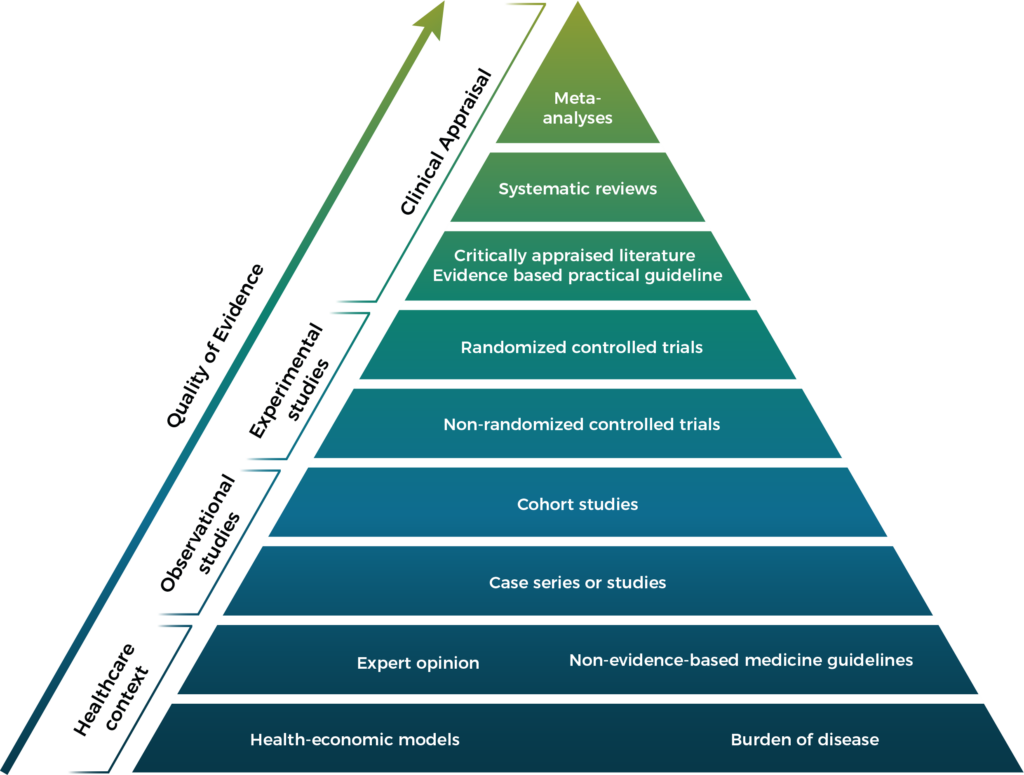
From May 2021 the new EU medical-device regulations are in force with a greater focus on high-quality evidence generation. Slowly, requirements for medical technologies are coming into line with those for pharmaceuticals. Medical devices, though, have unique challenges:
- Learning curve
- Incremental innovation
- Organizational impact
- Diversity
Read our publication on the health-economic landscape for medical device in Europe. Working with Coreva Scientific you will be supported by a team that knows your business. Where others may try and lever your product into the pharma scenarios that they know and trust, we build a solution for you.
Let us develop value for your project
We develop apps for
real-world evidence collection
Does published data meet your needs? Can registries and databases be used to assess your medical technology? Either way, we are here to help: If the answer is no, then we can develop apps to help capture the data you need to progress your product and its value story. If yes, we can analyze large data sets, extracting stories from data to improve your evidence base.
Working with
Coreva Scientific
We put a strong focus on communication and service. It is our belief that one phone call can replace and be more efficient than 20 emails. Want to meet face-to-face, we can be there. We have one requirement, mutual respect – on every project together we are partners. You are not a customer; we are not outsourcing. Together we build value.
Your product knowledge combined with our area expertise brings growth. All our research and analyses are scientifically and academically sound. Work with us safe in the knowledge that you can publish results in reputable journals and at top-line congresses.
You are kept up-to-date and your data is kept safe:
- Detailed project proposals
- Regular calls
- Full data-security compliance
- International, academic staff